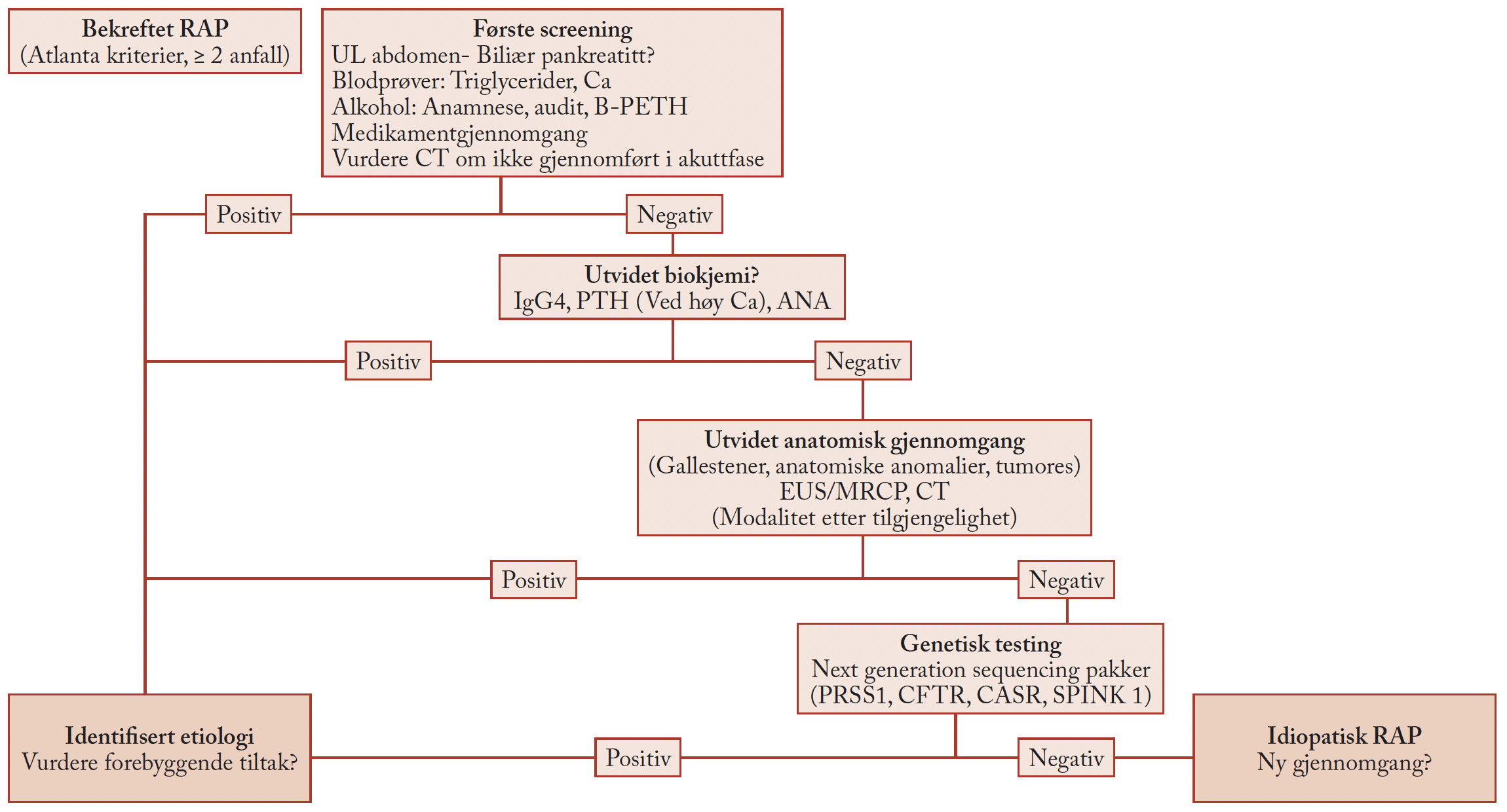
Tekst: Duan Chen. Department of Cancer Research and Molecular Medicine, Norwegian University of Science and Technology (NTNU), Trondheim.
The vagus nerve has been called the “wandering nerve” because of the many areas it innervates and because of the many diverse functions it has. The vagus nerve has two major components: efferent fibers and afferent fibers. The efferent fibers are associated with the autonomic nervous system from the brainstem to organs like the lungs, heart, and gut, whereas the afferent fibers carry sensory information from the organs back to the brainstem. Acetylcholine was originally described as “vagus stuff”, but now is known to be neurotransmitter at all autonomic ganglia, at many autonomically innervated organs, at the neuromuscular junction, and at many synapses.
Drug repurposing is an approach to find novel therapeutic indications for already approved drugs based on the mechanism-of-action (MOC). Thus, it goes usually directly to preclinical testing and clinical trials by taking the advantage of MOC. This approach has enabled us to test botulinum toxin A (Botox) vs. vagotomy for treatment of gastric cancer and Botox vs. “Vagal BLocking for Obesity Control” (VBLOC) for treatment of obesity.
Treatment of gastric cancer
It has become an interesting question for us whether vagus nerve promotes gastric cancer development, given that the vagus nerve-mediated acetylcholine muscarinic type 3 receptor (M3R) signaling potentiates a proliferative effect on gastric epithelium. In fact, most of gastric cancers in patients arise from lesser curvature of the stomach, where more pronounce vagus nerve ending is distributed. Therefore, we have investigated the potential role of the vagus nerve in gastric tumorigenesis.
Using different mouse models of gastric cancer, including genetically-induced cancer mouse model, chemically-induced cancer mouse model and a Helicobacter pylori-infected mouse model, we have found that vagotomy attenuated tumor cell proliferation1.
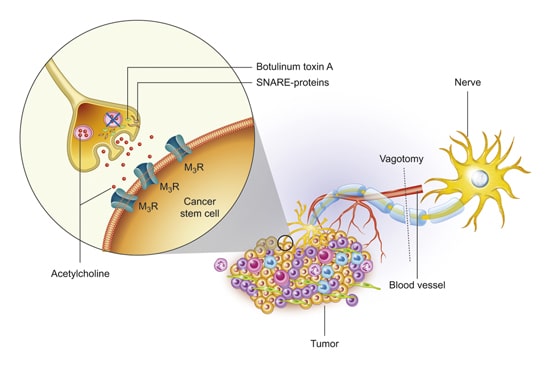
Drawing showing the action site (SNARE-proteins) of botulinum toxin A (Botox) in tumor, which is made based on the results of an original research article1.
Consistent with this finding, unilateral vagotomy or local injection of Botox in the gastric wall similarly impaired preneoplastic growth1 should be superscript. Vagotomy, Botox injection, or systemic treatment with darifenacin (M3R antagonist) given at later stages of gastric tumorigenesis also suppressed the tumor progression and augmented the anti-tumor effect of chemotherapy, leading to a prolonged survival1. This anti-tumor effect was mediated predominantly by inhibition of WNT signaling which controls the expansion of leucine-rich repeat containing G protein-couple receptor 5-positive stem cells in the gastric mucosa via activation of M3R (Fig. 1)1. In patients with gastric cancer, WNT signaling was associated with neural pathways, and neuronal density was correlated with more advanced tumors1. Thus, we have suggested that nerves is an important part of the stem cell niche and tumor microenvironment, pointing to the potential utility of anti-neurogenic therapies, such as local injection of Botox1.
Because the local injection of Botox has been successfully tested in animal models of gastric cancer1, a human clinical trial (phase II) using Botox through a conventional upper endoscopic approach for treatment of gastric cancer has been initiated at St. Olav’s Hospital in Trondheim (https://clinicaltrials.gov/ct2/show/NCT01822210). The implication of local injection of Botox for other GI cancers has not been reported yet, but can be great potential, as it blocks the so-called “nerve-cancer cell cross-talk” by acting on the soluble N-ethylmaleimide-sensitive factor-attachment protein receptor (SNARE) proteins2, 3. In patients with adenocarcinoma of esophagogastric junction, cholangiocellular cancer, pancreatic cancer or rectal cancer, the neural infiltration/invasion in the tumor is often observed and associated with poor prognosis4.

The research group at NTNU-St. Olavs Hospital: Standing from left-to-right: Steinar Lundgren, Jon Erik Grønbeck, Bård Kulseng, Ronald Mårvik and Gjermund Johnsen. Seated left-to-right: Magnus Kringstad Olsen, Helene Johannessen, Duan Chen, Chun-Mei Zhao and Gøran Andersen.
Treatment of obesity
The role of vagus nerve in physiologically controlling food intake and body weight has been studied during the past decades. It is believed that food interacts with the gut to provide the brain via vagus nerve afferents with information regarding food composition, amount of ingested food and energy content. The brain determines the rate of nutrient absorption, partitioning, storage, and mobilization through vagus nerve efferents as well as the sympathetic nervous system and hormonal mechanisms. This brain-gut axis is considered as an autonomic neurohumoral pathway regulating energy homeostasis. Previously, truncal vagotomy had been tried for treatment of severe obesity without great success. Recently, an electrical device, the so-called “Vagal BLocking for Obesity Control” (VBLOC), has been developed to block the subdiaphragmatic truncal vagus nerve with high frequency and low power electrical signals. This device can be implanted through the laparoscopy, and has been recently approved by the U.S. Food and Drug Administration to treat obese adults. The treatment concept is that it targets the vagus nerve pathway in the brain-gut axis to control hunger.
Utilizing different animal models, we have investigated the effects of VBLOC vs. Botox injection into the gastric wall in the region of antrum. Total body weight loss was 10% and >20% by VBLOC and Botox, respectively. One hundred twenty-five normal and high-fat diet-induced obese rats have been tested and no mortality, adverse effects and pathological changes were found in the animals subjected to Botox injection. We have found that Botox increased satiety ratio, leading to reduced food intake, and increased energy expenditure. Botox also reduced fasting blood glucose levels, while gastric emptying rate was unchanged. Gene expression in hypothalamus and the plasma levels of gut hormones were unchanged shortly after Botox injection. But later, the gene expression for neuropeptide Y and agouti-related peptide was increased, while pro-opiomelanocortin was decreased in arcuate nucleus in the hypothalamus, and the plasma levels of cholecystokinin, gastrin, and peptide YY, but not glucagon, glucagon-like peptide-1, were reduced, suggesting compensatory changes in the gut hormones-hypothalamus pathway5.
Because this preclinical trial has demonstrated the safety and efficacy of gastric injection of Botox, a human clinical trial (Phase II) using Botox through a conventional upper endoscopic approach for treatment of obesity has been currently conducted at St. Olavs Hospital in Trondheim
(https://clinicaltrials.gov/ct2/show/NCT02035397).
References:
- Zhao CM, Hayakawa Y, Kodama Y, et al. Denervation suppresses gastric tumorigenesis. Sci Transl Med 2014;6:250ra115.
- Jobling P, Pundavela J, Oliveira SM, et al. Nerve-Cancer Cell Cross-talk: A novel promoter of tumor progression. Cancer Res 2015;75:1777-1781.
- Meng J, Wang J. Role of SNARE proteins in tumorigenesis and their potential as targets for novel anti-cancer therapeutics. Biochim Biophys Acta 2015;1856:1-12.
- Liebl F, Demir IE, Mayer K, et al. The impact of neural invasion severity in gastrointestinal malignancies: a clinicopathological study. Ann Surg 2014;260:900-7.
- Johannessen H, , Olsen MK, et al. Preclinical trial of gastric injection of botulinum toxin type A as weight-loss-surgery. Gastroenterology 2014;146:S-1077.